Abstract
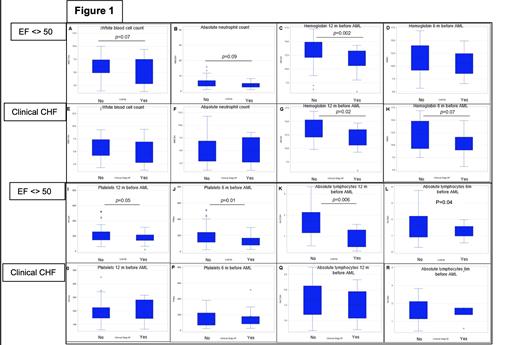
Background: There is strong association between clonal hematopoiesis of undetermined prognosis (CHIP) and coronary artery disease (CAD) development. Indeed, ischemic heart disease is a leading cause of congestive heart failure (CHF) in western society. CH induces not only enhanced risk for myeloid malignancies, but also significant cardiovascular morbidity in elderly patients (pt). Pt with impaired ejection fraction (EF) and clinical manifestation of heart failure frequently initiates systemic pro-inflammatory state [SPS] characterized by interleukin-6 (IL6) and tumor necrosis factor alpha (TNF-α) upregulation. Previous studies suggest that such an inflammatory state is capable of sustaining and/or accelerating hemopoietic clonal selection/dominance. How these CHF induced inflammatory changes affect "peripheral blood cell count composition' in pt with suspected CHIP is unknown. However, modifications in peripheral blood count data "months before hemopoietic malignancy diagnosis" may inform association between clonal dynamics and hemopoietic output under CHF stress. The primary objective of our study was to investigate effect of heart failure in "pre- AML diagnosis" peripheral blood count data to detect "early" hemopoietic modifications induced by CHF. Methods: After IRB approval, AML pt diagnosed with and without CHF were selected for analysis. Given possible differential inflammatory effect between pt exhibiting CHF with and with low EF, we analyzed peripheral blood cell count in pt with "echocardiographically confirmed" EF <> 50% and those pt with and without clinically documented CHF [cdCHF]. Descriptive statistics was performed for categorical and continues variable with Chi-square and t-test. SAS software was used for analysis. Results: 27/152 (17.7%) and 125/152 (82.2%) pt developed or not EF <50%. Median age was 71.1 years (y) v 61.2 y, in pt with and without EF<50%, p=<0.0001. Aging was negatively correlated with EF, [R= -0.25, p=0.001]. Median age was 73 y v 61 y, for pt with and without cdCHF, p=0.001. 100% and 82% of pt were male among those with and without EF <50%. In pt who had or did not have EF <55% Favorable (fav), Intermediate (Int) and unfavorable (unfav) ELN-2017 subgroups were 3.23% vs 13.6%; 42% vs 46%; and 55% vs 40%, p=0.07. To address our main hypothesis that CHF induces "early pre-diagnostic AML' hemopoietic modifications, CBC data at 6 and 12 months (m) before AML onset in pt with and without EF<50% v those with and without cdCHF was extracted. At 12 m prior AML diagnosis, EF<> 50% detected differential expression for WBC [p=0.07], Hemoglobin [p=0.002], platelets [p=0.05] and absolute lymphocyte count (ALC) [p=0.006] [Fig 1, panel A, C, I, K]. Similarly, EF <>50% at 6 m prior AML diagnosis detected differential expression for platelets [p=0.01], ALC [p=0.04] [Fig 1, panel D, J, L]. In contrast, cdCHF detected only differential hemoglobin at 12 m [p=0.01]. Conclusions: CHF induces quantitative count defects 6 and 12 m before AML diagnosis. Changes are characterized by WBC, hemoglobin, platelets and ALC decline that are directly correlated with objective left ventricular EF impairment, but not with only clinical CHF. Our study adds body of evidence to support the role of CHF with low EF as "hemopoietic cell extrinsic stressor'. CHF associated with low EF seems to be prerequisite for hemopoietic stress in hosts with already initiated clonal hematopoiesis. Our findings have important experimental implications, especially for studies that seek to understand how cell extrinsic stressors facilitate clonal progression.
No relevant conflicts of interest to declare.